Submission and other forms Tests, fees and user guide Client web portal Contacts Online payments
ADRDL will be implementing a new password policy on May 15th.
Update your password by logging into your ADRDL Client Web Portal https://adrdl.sdstate.edu/login.php
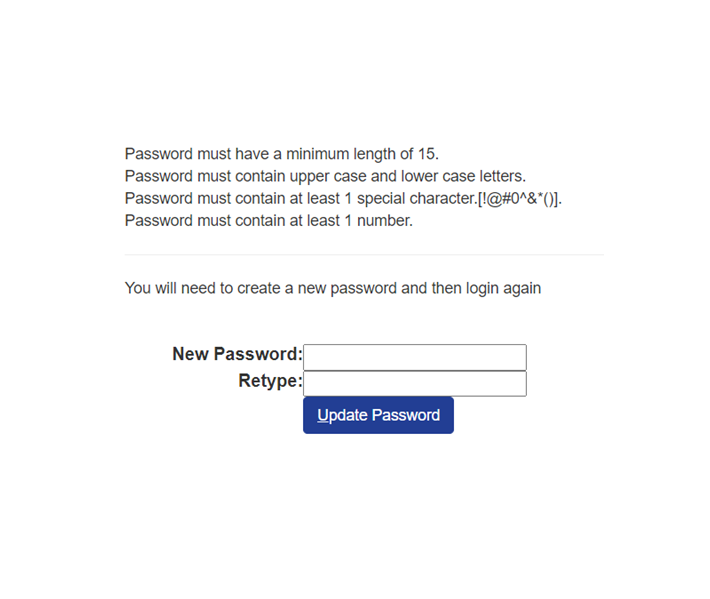
Password must have a minimum length of 15.
Password must contain upper case and lower case letters.
Password must contain at least 1 special character. [!@#^&()]
Password Must contain at least 1 number
You will need to create a new password and then login again.
Update Profile ---- new password:
https://adrdl.sdstate.edu/formUpdateProfile.php
Questions? Contact ADRDL IT:
sdsu.adrdlit@sdstate.edu
Changes to Rabies Testing at SD ADRDL
Rabies testing – changes to billing procedures.
Please note the following changes to the billing for animal rabies testing specimens sent to the South Dakota Animal Disease Research and Diagnostic Laboratory (ADRDL).
Human Exposure to Wildlife
Effective Sept. 1, 2023, South Dakota Game, Fish and Parks will only pay testing fees for wildlife animal species that have posed a risk of human exposure to rabies. This includes live wildlife species that have bitten, scratched, or otherwise exposed a person or persons. In the past, Game, Fish and Parks had paid testing fees for all wildlife species, regardless of human exposure risk.
Human Exposure to Domestic Animals
The South Dakota Department of Health will continue to pay testing fees for domestic animal species that have posed a risk of human exposure to rabies.
Animal-only or No Exposure
Specimens for rabies testing animals that have exposed other animals, or have not exposed either people or animals, will be billed to the submitting clinic, regardless of whether the specimen comes from a wildlife or domestic animal species.
Prior to testing, veterinarians and submitters need to fill out the ADRDL rabies submission form (https://www.sdstate.edu/sites/default/files/file-archive/2019-05/Rabies-Diagnostic-Requisition-Form.pdf) completely and with appropriate detail about human exposures. Information from this form is used to contact people with possible exposures and ensure they get prompt, appropriate treatment should the specimen test positive for rabies. Supplying sufficient detail on the submission form also ensures that testing fees are appropriately charged.
Please call the ADRDL at 605-688-5171 with questions.
South Dakota Animal Disease Research and Diagnostic Laboratory
South Dakota Game Fish and Parks
South Dakota Department of Health
South Dakota Animal Industry Board

SDSU Undergraduate Students Learning in New Anatomy Lab
SDSU Undergraduate Students Learning in New Anatomy Lab.
Over 120 SDSU students taking Anatomy and Physiology of Domestic Animals are already enjoying the learning experiences in the newly renovated anatomy lab space at the Animal Disease Research and Diagnostic Laboratory (ADRDL).
The new space is part of the renovated ADRDL facility completed this past summer. With the move of diagnostic services to the new addition to the north, the former ADRDL was turned into labs and classrooms for teaching in the upcoming Professional Program in Veterinary Medicine ("2+2" program) and undergraduate courses, in addition to research work.
The new anatomy lab occupies the former ADRDL necropsy room. It features the very latest in instructional technology, with cameras that can zoom in on anatomic specimens on two different tables and project onto a large video screen. New downdraft dissection tables (bottom picture above) have been installed, allowing safe and comfortable work with cadavers and other materials. Faculty members are grateful to the University of Minnesota College of Veterinary Medicine for loaning some additional specimens for veterinary anatomy.
In addition, the room will have a research function, being available for use as a necropsy floor for research projects.
Anatomy and Physiology of Domestic Animals (known as "A and P" to students past and present) is a requirement for animal science and dairy science majors, as well as a required course for students completing the Animal Health minor. Drs. Christina Larson (lectures) and Bev Cassady (labs) are the instructors.

Dr. Tamer Sharafeldin Joins ADRDL as New Pathologist; Brings Poultry Expertise
Dr. Tamer Sharafeldin is the ADRDL's new pathologist, beginning his work at SDSU on June 1. Dr. Sharafeldin received his professional pathology training and his Ph.D. at the University of Minnesota. While his work will include all the routine casework coming through the diagnostic lab, Dr. Sharafeldin brings a particular expertise in poultry diseases to SDSU. He has wide experience with a variety of poultry production systems including turkey and layer production as well as game birds in Minnesota and Pennsylvania. He is one of the pioneers who studied the pathogenesis and the immune response of turkey arthritis/tenosynovitis reovirus. He has a nationally recognized turkey reovirus research program that received research grants to study and develop turkey reovirus vaccines.
Tamer is working with colleagues at SDSU to expand poultry diagnostics in support of turkey and layer production systems in the upper Midwest. "The ADRDL has an already-strong reputation for diagnostics development. By submitting poultry cases to SDSU you're not only benefiting from diagnostic service from an experienced avian pathologist, but you're also helping support the development of more diagnostics here at SDSU."
Contact Dr. Sharafeldin with questions at 605-688-5171.
Approach to COVID-19 Similar to Battling Swine Pathogens
One of the most important things to understand is the concept of herd immunity. Individual animals can become immune by recovering from an earlier infection or through vaccination. Some animals cannot become immune due to their age, stress (weaning, environmental conditions), co-infections and for this group herd immunity is a crucial method of protection.
Once a certain threshold of the population is immune, herd immunity gradually eliminates a disease from a population. The term "herd immunity" implies that it must have been developed in livestock but actually it was a term first developed in the 1930s to describe a phenomena observed after a significant number of children had become infected and immune to measles, the number of new infections temporarily decreased, including among susceptible children. It was soon realized. Read the article by Dr. Chris Chase (National Hog Farmer).

South Dakota ADRDL Fulfills Need for Human COVID-19 Testing
South Dakota State University students can know within 24 to 48 hours whether they are positive for COVID-19, thanks to Animal Disease Research and Diagnostic Laboratory on campus at the South Dakota Animal Disease Research and Diagnostic Laboratory on campus.
ADRDL director Jane Christopher Hennings said, “Diagnosing COVID-19-positive individuals as soon as possible is an essential part of slowing the virus spread.” ADRDL, which has both biosafety level 2 and 3 laboratories, is one of more than 20 laboratories in the U.S. Department of Agriculture National Animal Health Laboratory Network certified to do human testing to combat the COVID-19 pandemic.
The One Health Laboratory uses the polymerase chain reaction, or PCR, test, the same test the South Dakota Department of Health performs to diagnose COVID-19. Thus far, One Health has met COVID-19 testing needs for SDSU’s Student Health Clinic and Counseling Services, but the laboratory is available to support the South Dakota Department of Health’s testing efforts, if needed.
“Our scientists run about 200,000 PCR tests every year on animals alone, so we are used to doing this type of high complexity, high throughput testing,” Hennings said. ADRDL scientists have played a key role in diagnosing animal disease outbreaks, such as the highly pathogenic avian influenza and porcine coronaviruses.

BVDV Persistent Infection, Novel Bosavirus Characterized in Bison through SDSU Work
As part of an investigation of reproductive losses in an American bison herd, faculty and staff at SDSU's ADRDL characterized for the first time persistent infection with Bovine Viral Diarrhea Virus (BVDV), as well as a novel bosavirus in bison.
Following reproductive problems in a bison herd during the 2018-19 calving season, investigators collected samples from the breeding herd as well as 4 animals with failure to thrive. Serology, virus isolation, metagenomic sequencing and pathology was performed.
All 26 animals examined serologically had titers to BVDV Type 1 (range 1:512 to 1:8192, with 17 with titers greater than 1:1000) and BVDV Type 2 (range 1:64 to 1:8192, with 7 greater than 1:1000), despite the lack of recent BVDV vaccination. Metagenomic sequencing on pooled nasal swabs and serum identified co-infection of BVDV and bovine bosavirus. The BVDV genome was most similar to the BVDV type 1a vaccine strain Oregon C24V with 92.7% identity in the coding region. Bosavirus was also identified but its clinical significance is unknown.
Pathology examination did not reveal any gross lesions. On histopathology, two BVDV positive animals had lymphoid depletion in the ileo-cecal valve lymphoid region. A female PI bison had a decrease in primary follicles in the ovary, and a male PI bison showed evidence of decreased spermatogenesis in the testes.
Serum from these same animals collected two months later remained positive for BVDV and bosavirus, with one animal co-infected with both BVDV and bosavirus. These results suggest that both viruses can persistently infect bison. While the etiological significance of bosavirus infection is unknown, the ability of BVDV to persistently infect bison has implications for BVDV control and eradication programs.
Investigators from SDSU on the project included Drs. Angela Pillatzki, Ben Hause and Chris Chase. They can be contacted at 605-688-5171 for more information.

Drs. Moisan and Siddiqui Join VBSD and PPVM as New Faculty Members
Two veterinarians have joined SDSU's Veterinary and Biomedical Sciences Department, playing key roles in educating students in the Professional Program in Veterinary Medicine ("2+2" Program).
Dr. Peter Moisan (top picture) will be teaching General Pathology and Systemic Pathology. A Canadian citizen, Dr. Moisan grew up in Tennessee, achieving his DVM from the University of Tennessee in 1981. He also holds an M.S. degree in Microbiology, and has spent much of his career working with infectious diseases of domestic animals, particularly cattle and swine.
After veterinary school, Dr. Moisan spent 11 years in cattle and swine practice in Manitoba and Alberta before entering the Pathology Program at Michigan State University. While studying for Pathology Boards, he also became Board Certified in Food Animal Practice and Beef Practice with the American Board of Veterinary Practitioners. His professional experience includes Pasteurella multocida research at Kansas State, 21 years as a pathologist at the Rollins Diagnostic Laboratory in Raleigh, NC, work for Antech Diagnostics and pathology work at the New Mexico veterinary diagnostic laboratory before coming to SDSU. Dr. Moisan is also an adjunct professor at the North Carolina State University College of Veterinary Medicine.
Dr. Aziz Siddiqui (bottom picture) will coordinate and teach Integrated Biochemistry and Physiology in the 2+2 program, and contribute to other courses as well. A native of Bangladesh, Dr. Siddiqui has DVM, M.S., and Ph.D. degrees from Bangladesh Agricultural University, specializing in veterinary obstetrics and theriogenology. In 2002, he moved to the University of Wisconsin as a USDA exchange research scholar, eventually completing post-doctoral training in endocrinology and reproductive physiology at the University of Wisconsin School of Veterinary Medicine.
Before coming to SDSU, Dr. Siddiqui taught anatomy and physiology, and performing research on gamete and embryology at multiple campuses within the University of Wisconsin system. Prior to that he was the Andrology Laboratory Director at Accelerated Genetics – a large bull stud in Westby, Wisconsin. Dr. Siddiqui's specialty is in ruminant theriogenology, with a research interest in andrology and embryology where he uses ultrasonography as the primary clinical/research tool.
Welcome Drs. Moisan and Siddiqui to SDSU!

Sarah Vos Joins 2+2 Program as Coordinator for Recruitment and Academic Services
A key role within SDSU's Professional Program in Veterinary Medicine ("2+2" Program) is now filled with the hiring of Sarah Vos. Sarah's responsibilities in this role include recruiting students to the program, coordinating student events and activities and managing marketing and promotional material, including social media and websites.
A native of Rapid City, Sarah graduated from SDSU in May 2021 with a B.S. degree in Animal Science, with minors in Animal Health, Equine Studies and Biology. While at SDSU, Sarah competed on the SDSU Rodeo team in goat tying and breakaway roping, along with serving as secretary for the team. She also served as a College of Agriculture, Food and Environmental Sciences ambassador and a teaching assistant for multiple courses. Sarah's experiences with the VBS department during undergrad also run deep, as she completed an independent study research project, “Bosavirus in Bison: Determining tissue trophism in normal and infected animals” under Dr. Chris Chase.
Much of Sarah's spare time is spent with her three horses and her Border Collie/Australian Shepherd, Lucy.
"I am so excited to be back at SDSU and working with this amazing new program, where my responsibilities are always evolving," says Sarah.
Welcome, Sarah, back to SDSU!

Food Safety Lab Gains "Same As" Status, Adds Salmonella enteritidis Test
Recent additional accreditation by the ADRDL's Food Safety Microbiology Lab is contributing to an easier path for South Dakota's meat processors to ship their products across state lines.
In June 2021, the South Dakota Meat Inspection Program, led by the state's Animal Industry Board (AIB), entered into an agreement with the USDA/FSIS to enter the Cooperative Interstate Shipment Program (CIS). Under CIS, state-inspected plants can operate as federally-inspected facilities, under specific conditions and ship their products in interstate commerce and internationally.
To be part of this agreement, the AIB needed to demonstrate their inspection procedures and policies are the "same as" those utilized by the USDA. This includes laboratory testing.
Faculty and staff with the Food Safety Microbiology Lab worked to gain ISO 17025:2017 accreditation for test procedures for Salmonella, Listeria monocytogenes, E. coli O157:H7, and non-O157 Shiga toxin-producing E. coli (STEC). This higher level of accreditation mirrors that of USDA/FSIS labs in California, Missouri and Georgia, and puts SDSU's lab in the same class as just a handful of other state labs.
Additionally, the lab is now offering Salmonella enteritidis (SE)-specific testing. The test process used is the Romer Labs RapidChek SELECT™ Salmonella enteritidis Test System, which utilizes highly selective media and lateral flow strips to screen for SE. Positive samples are confirmed via immunomagnetic separation (IMS) and rapid serotyping rule-out at NVSL. The test has been validated on drag swabs, egg pools and chicken carcass rinses, is compliant with the NPIP program and is a test method equivalent for the FDA SE Egg Safety Rule (21 CFR Parts 16 and 118). Lab staff will run samples that arrive Monday – Wednesday with negative results available in 48 hours.
Food Safety Microbiology Lab staff include section leader Dr. Joy Scaria, research associate Laura Ruesch, and senior microbiologists Kara Hendrickson-Guttum and Zach Lau.

SDSU Welcomes Veterinary Students with Unique Blue Coat Ceremony
SDSU Welcomes Veterinary Students with Unique Blue Coat Ceremony
Students comprising the first class of the SDSU professional program in veterinary medicine were honored at the Inaugural Blue Coat Ceremony November 5, where the students were presented with a personalized blue Carhartt work coat.
“We carefully selected our first class of students. All of them are exceptionally bright and committed to learning the many aspects of veterinary medicine,” said Gary Gackstetter, professor and director of the professional DVM program in veterinary medicine. “We are confident they will all contribute to both animal and public health across the upper Midwest. These new blue coats are also a symbol of our confidence in these students.”
The coats presented to each student represent not only the hard work they have completed to get to this point in their education, but the hard work ahead of them as they continue their education in veterinary medicine.
“The blue Carhartt jacket is symbolic of the hard work and dedication that the professional program in veterinary medicine will require,” said Jessica Juarez, assistant professor and assistant director of PPVM. “It is a way for our students to proudly represent SDSU and each other when out on internships and other opportunities where they can wear the jacket.”
Each coat presented at the ceremony was sponsored by a local veterinarian, symbolic of the support these students can expect.
Reduced Pricing for Shipping
NOTE: Starting April 1, as per the AAVLD Accreditation requirements, full diagnostic laboratory reports will be sent labeled as "preliminary", indicating some results are still pending or "final", where no test results are pending. Previously, for some cases, only the newest results were sent via e-mail or fax.
Thank you for understanding this requirement. If you have any questions, please feel free to contact the laboratory.


